Reporting forms and information can be found at https://yellowcard.mhra.gov.uk/.
Adverse events should also be reported to Luye Pharma Ltd at safety@luyepharma.co.uk.
To view the Prescribing Information click here
Reporting forms and information can be found at https://yellowcard.mhra.gov.uk/.
Adverse events should also be reported to Luye Pharma Ltd at safety@luyepharma.co.uk.



8 Zeyzelf® patches

8 Adhesive covers

Patient information leaflet (PIL)
*The adhesive cover can be written on to remind patients/carers when the patch was applied and/or when it should be removed.
Adverse events should be reported.
Reporting forms and information can be found at https://yellowcard.mhra.gov.uk/
Adverse events should also be reported to safety@luyepharma.co.uk
For prescribing information scan the QR code:

Rivastigmine is a recommended AChE (acetylcholinesterase) inhibitor for managing mild to moderate Alzheimer’s disease.
When prescribing an AChE, treatment should usually be started with the drug with the lowest acquisition cost. However, an alternative AChE inhibitor may be prescribed if it is more suitable, considering factors like side effects, expectations about adherence, medical comorbidity, potential drug interactions and dosing schedules.

Download the Patient Leaflet here
This link will take you to the electronic Medicines Compendium (eMC) website.
Download information on how to use Zeyzelf® and
the patient diary here
This link will take you to the electronic Medicines Compendium (eMC) website.
Download the SmPC
This link will take you to the electronic Medicines Compendium (eMC) website.
TECHNICAL INFORMATION
Active Ingredients:
Rivastigmine
Therapeutic area:
Neurology
Pharmaceutical Form:
Transdermal Patch
Prescribing Zeyzelf® by brand is less expensive than prescribing rivastigmine generically, costing £35.09 for a 28-day supply compared to £77.9720 Zeyzelf®‘s manufacturer, Luye Pharma, produces all it’s transdermal products in its dedicated European production facility, providing greater oversight of the entire supply chain and reliability for clinicians, pharmacists and patients.
To ensure patients receive Zeyzelf® twice weekly rivastigmine patches and to reduce the risks of dispensing errors, it is important that prescriptions are written by brand.21
Following the discontinuation of Alzest transdermal patches,1 Zeyzelf® twice weekly rivastigmine patches is competitively priced and is more the 50% less expensive than the originator single day patch, Exelon. List prices costs of other available single day rivastigmine patches are shown below for comparison. Zeyzelf® are now the reliable choice for managing your patients.
*8 patches of Zeyzelf® provides 28 days treatment
The price comparison is based on Zeyzelf® 28 days verses Alzest (discontinued) and Almuriva at 30 day.
Lower risk of
overdose5
preferred transdermal patches to capsules overall*. Carers preferred the patch over capsules for the ease of use and following the schedule. Carers also reported greater satisfaction and less interference with daily life with patches compared with the capsules. 6
Thus, the simplification of treatment regimens may assist in reducing the burden on caregivers, which may subsequently result in enhanced medication adherence and a positive impact on the patient’s condition.
At 24 weeks, patient compliance was significantly better with the twice weekly patch than with the daily patch(p=0.002). Caregiver burden significantly improved at the end of the treatment.
Seventy point seven (70.7%) caregivers at week 12 and 87 (94.6%) caregivers at week 24 reported that the patient always took the twice weekly patch as prescribed versus 27 (29.3%) of patients taking the daily patch.
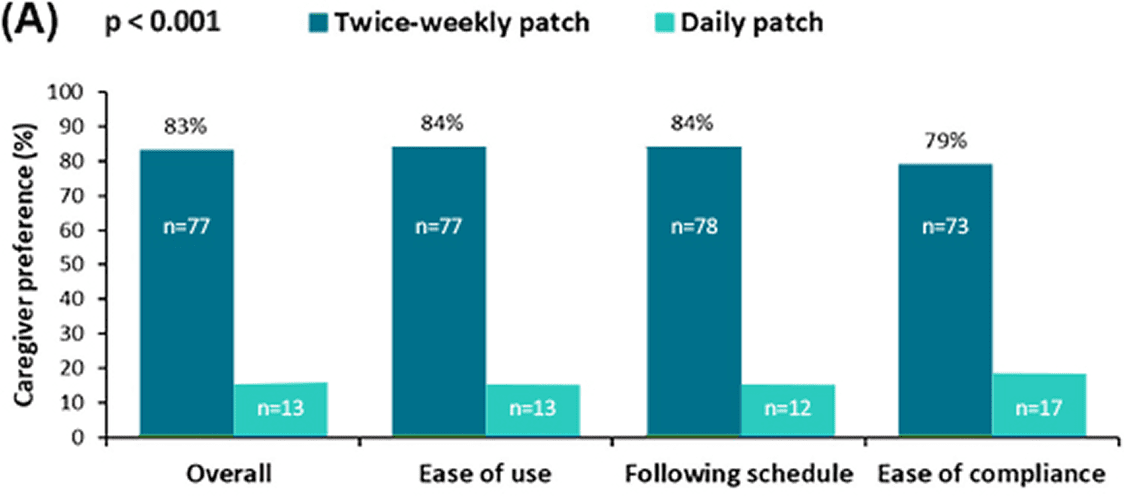
Figure 1. Caregiver preference and satisfaction and compliance with treatment. (A) Caregiver preference and reasons provided for preference for twice weekly or daily rivastigmine patch at 24 weeks (n=90). For all outcomes, p < 0.001, twice weekly patch versus daily patch.
A study comparing the twice weekly Zeyzelf® patch and Exelon® daily patch showed Zeyzelf® had better adhesion properties despite the longer dosing intervals.8
Percentage of assessments showing ≥90% adhesion for Zeyzelf® compared to <70% for Exelon®.8
Confidence Interval (CI) of mean adhesion above 90%.

Dysphagia (difficulty swallowing) is common among older adults with AD and can make it challenging for patients to adhere to oral AChE inhibitor treatments. Transdermal patches such as Zeyzelf® offer a non-oral option for patients who may have trouble swallowing pills, ensures consistent delivery of medication and may reduce the risk of missed doses due to swallowing issues.10
Unlike other AChE inhibitors, rivastigmine demonstrates a low potential for drug-drug interactions, making it an option for patients on polypharmacy medications.1 Bypassing the first-pass metabolism in the liver, the patch further reduces the risk of interactions with other drugs.
Transdermal administration delivers reduction in certain side effects vs oral rivastigmine due to:13,14
If adverse events occur, drug delivery can be promptly ceased by simple patch removal.14
In addition to lower risk of side effects, transdermal route of administration can provide greater adherence to the treatment regimen and has less risk of dose dumping compared with the oral route. This is especially important in older patients who may need concomitant treatment for multiple conditions.15
A rivastigmine 9.5mg/24hr transdermal patch provides efficacy similar to the highest recommended daily dose of oral rivastigmine (6 mg bid) with improvements in dementia symptoms including:4, 8
Fewer people using Zeyzelf® reported nausea, vomiting, or diarrhoea compared to those taking oral medication.13 The rivastigmine transdermal patch tends to have a favourable side effect profile, particularly regarding gastrointestinal effects and overall tolerability. Zeyzelf®’s transdermal delivery system generally has a lower propensity to cause side effects most commonly associated with oral AChE inhibitors from 1 in 10 to 1 in 100.
As Zeyzelf® is a patch, not a pill, it means it avoids the stomach and the digestive system. This makes it more tolerable and is particularly beneficial for older patients who may have a sensitive stomach or suffer from Gastrointestinal issues (GI). Fewer people using Zeyzelf® reported nausea, vomiting, or diarrhoea compared to those taking oral medication. Skin reactions are usually mild, and the patch has been designed to stick well with less irritation as its only applied only twice a week versus the daily patch.1

Security and peace of mind

Skin adhesion is one of the most important functional properties for a transdermal patch and is critical to the tolerability, efficacy and quality of the patch.17
Zeyzelf® twice weekly patches come with an oval shaped adhesive cover to optimise adherence to the skin. In a study comparing the twice weekly Zeyzelf® patch and the Exelon daily patch, Zeyzelf® demonstrated superior adhesion properties compared to the Exelon daily patch, despite the longer dosing interval.8
Zeyzelf® twice weekly patches offer a unique and simple solution to improve medication adherence in Alzheimer’s patients:
Visual marker: Helping minimise
the risk of multiple dosing errors
Direct Notation: The adhesive cover can be written on with the date of application and/or the date it should be removed helping carers record and track the medication
The Zeyzelf® twice weekly patch with its adhesive cover provides security with peace of mind for the patients and their care givers.

Less frequent application vs a daily patch gives the option of dosing (application) free days for the carer and patients, e.g., weekends could be dosing (application) free if the patches are applied on a Thursday and Monday or Friday and Tuesday.
Zeyzelf® is applied on fixed days of the week. Establishing fixed days of application will help the patient and caregiver to maintain a routine and adherence to the medication.2
With the ease of fixed day twice weekly dosing, carers can more easily plan the dosing schedule to the patient’s and their own convenience.1
Example schematic representation comparing patch application of Zeyzelf® to a daily patch.

Helping to simplify the lives of patients as well as their caregivers with:
For information on Instructions for use including medication record sheets for patients that have been prescribed Zeyzelf® Patch (rivastigmine transdermal system) to ensure the correct use of the patch please scan the QR code
Or visit:
https://www.medicines.org.uk/emc /product/14957/rmms

Patient Diary: Please scan the QR code for information for use and a patient diary or visit: https://www.medicines.org.uk/emc /product/14957/rmms

Additional security: with peace of mind for patients and caregivers

Adverse events should be reported.
Reporting forms and information can be found at https://yellowcard.mhra.gov.uk/
Adverse events should also be reported to safety@luyepharma.co.uk
For prescribing information scan the QR code:

References
1. Zeyzelf® rivastigmine transdermal patch, Summary of Product Characteristics, Luye Pharma Ltd. https://www.medicines.org.uk/emc/product/14957/smpc#gref. (Last accessed December 2025).
2. NICE guideline [NG97] June 2018. Available at: https://www.nice.org.uk/guidance/ng97. (Last accessed December 2025).
3. García-Alberca JM, De La Guía P, Gris E, et al. Journal of Alzheimer’s Disease. 2024;102(3). https://journals.sagepub.com/doi/abs/10.1177/13872877241292018 (Last accessed December 2025).
4. Piñol Ripoll G, Salas Carrillo M. Patient Prefer and Adherence. https://doi.org/10.2147/PPA.S510634. (Last accessed December 2025).
5. Poison: https://www.poison.org/articles/donepezil-can-be-toxic-if-too-much-is-taken-or-child-swallows-174. (Last accessed December 2025).
6. Blesa R, et al. Neurology. 2007;69(4 suppl 1):S23–S28. https://pubmed.ncbi.nlm.nih.gov/17646620/. (Last accessed December 2025).
7. Bullock R, et al. Curr Med Res Opin. 2005;21(8):1317–1327. https://pubmed.ncbi.nlm.nih.gov/16083542/. (Last accessed December 2025).
8. Schurad B, et al. Current Alzheimer Research. 2022;19,541–553. https://pubmed.ncbi.nlm.nih.gov/36017827/. (Last accessed December 2025).
9. Farlow MR. Int J Clin Pract Suppl. 2002127):37;(–44. https://pubmed.ncbi.nlm.nih.gov/12139366/. (Last accessed December 2025).
10. Parsons C. Withdrawal of Antidementia Drugs in Older People: Who, When and How? Drugs Aging 2016; 33:545-556. https://pubmed.ncbi.nlm.nih.gov/27393698/. (Last accessed December 2025).
11. Guener M, et al. BMC Geriatrics. 2023;23:304. https://pubmed.ncbi.nlm.nih.gov/37198547/. (Last accessed December 2025).
12. Grossberg GT, et al. Int J Geriatr Psychiatry. 2000;15(3):242–247. https://onlinelibrary.wiley.com/doi/10.1002/(SICI)1099-1166(200003)15:3%3C242::AID-GPS110%3E3.0.CO;2-7. (Last accessed December 2025).
13. Winblad B, et al. Int J Geriatr Psychiatry. 2007;69(4 Suppl 1):S14–22. https://pubmed.ncbi.nlm.nih.gov/17646619/. (Last accessed December 2025).
14. Sozio P, et al. Neuropsychiatr Dis Treat. 2012;8:361–368. https://pmc.ncbi.nlm.nih.gov/articles/PMC3428243/. (Last accessed December 2025).
15. Vadivelu N, Hines R L. Clin Interv Aging. 2008;3(3):421–430. https://pmc.ncbi.nlm.nih.gov/articles/PMC2682375/. (Last accessed December 2025).
16. Electronic Medicines Compendium (EMC). Zeyzelf® SmPC. Available online at: https://www.medicines.org.uk/emc/product/14957/smpc. (Last accessed December 2025).
17. Wokovich A M et al. Eur J Pharm Biopharm. 2006;64(1):1–8. https://www.sciencedirect.com/science/article/abs/pii/S0939641106000804. (Last accessed December 2025).
18. Comparative costs against non-tablet formulations table. Data on file.
19. Alzheimer’s Society. What are the costs of dementia care in the UK. Available online at: https://www.alzheimers.org.uk/about-us/policy-and-influencing/dementia-scale-impact-numbers. (Last accessed June 2025).
20. https://www.nhsbsa.nhs.uk/sites/default/files/2026-01/Drug%20Tariff%20Preface%20February%202026.pdf
21. BNF listing: https://bnf.nice.org.uk/drugs/rivastigmine/
You are now leaving the Luye Zeyzelf product website to go to
an external website over which Luye Pharma has no control
and to which our Privacy Policy does not apply. You are
solely responsible for your interactions with that website.
Click OK to proceed.
You are now leaving the Luye Zeyzelf product website
and will be redirected to our corporate site which
will open in a new window
Click OK to proceed.
You'll be redirected to:
UK-ZEY-105 April 2026
To receive the resource please provide your name, email address and NHS postal address below:
"*" indicates required fields
UK-ZEY-100ab April 2026
To view the resource please provide your name and email address:
"*" indicates required fields
UK-ZEY-127 April 2026